Home » Cancer Types » Prostate Cancer
Prostate Cancer
- Overview
- Treatment Options
- What to Expect
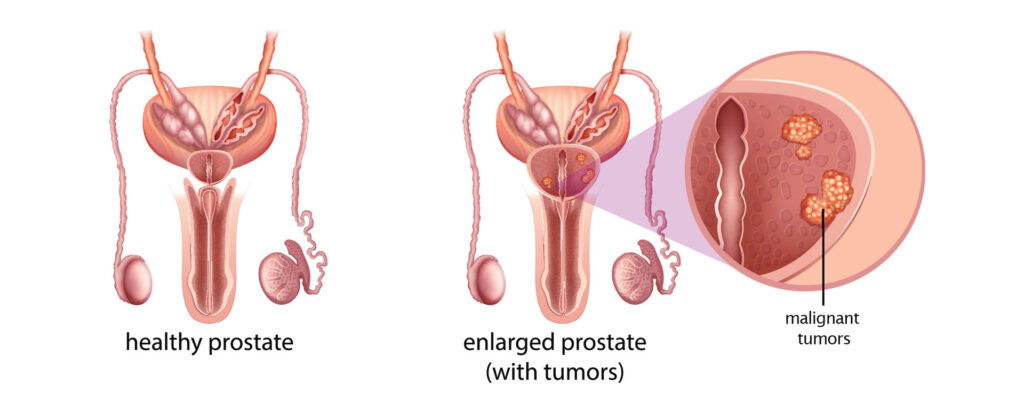
Prostate cancer is the one of the most common cancers in men in the United States. Approximately one in six men will develop prostate cancer according to the American Cancer Society. Prostate cancer tends to progress much more slowly than most other cancers and is generally associated with a good prognosis. Prostate cancer that has grown enough to bring attention to itself in the form of a lump or an elevated PSA has likely been present for at least a couple of years.
Surgery, radiation and hormone therapy are often the mainstays of treating prostate cancer. Some patients may derive the most benefit from a combination of treatment strategies. As such, our team of radiation oncologists, medical oncologists, and urologists will work with our patients to determine an optimal treatment strategy.
Watchful Waiting
In prostate cancer, it is usually not necessary to intervene at once. Many men who have prostate cancer are NOT going to die of it, and perhaps will never even be inconvenienced by it. If you have low risk disease, especially if your PSA is not rising rapidly and you don’t have a high percentage of positive biopsies, we may discuss deferring intervention for a time to see if there is any progression. This is even more advisable if you are older, or have other medical conditions that limit your life expectancy, which you can look up by clicking here. You can monitor the PSA and rectal exams at intervals and have annual prostate biopsies. You can choose to intervene in a few months, or a few years, if there is progression.
Hormone Therapy
Prostate cancer grows better in the presence of male hormones. Think of it as putting fertilizer on weeds: it doesn’t create the weeds, but it does make them grow better. If the fertilizer is withdrawn, the weeds will grow less well. Some of the weeds may even die for lack of fertilizer, though probably some will succeed in surviving. Dr. Charles Huggins received the Nobel Prize for his discovery, in the 1930’s, that men with advanced prostate cancer experienced dramatic improvement when they were castrated. In the modern day we rarely use actual castration, but drugs called LHRH Agonists have been developed to shut off the production of male hormones for up to several months after a single injection. Sometimes, additional drugs called anti-androgens are given in conjunction with the LHRH agonist. This is called “combined androgen blockade” but it is still a matter of some controversy as to whether it is superior to the LHRH agonist alone. As alluded to above, this is not a cure for prostate cancer, but it can be a remarkably effective treatment, sometimes able to control the progression of the disease for years, or even decades. It is an excellent choice for a man who already has evidence of spread, or a man who has very high risk disease or a limited life expectancy.
Hormone therapy is also often used in conjunction with radiation therapy, especially for patients with high risk disease. It appears to help in making the radiation therapy more effective locally, and also prevents or delays the appearance of cancer that may already have spread.
Blocking the male hormones has predictable side effects. Almost all men find that their sexual desire diminishes, as does their ability to have erections. Most men have hot flashes, similar to a woman going through menopause. These can range from temporary and mild to prolonged and very troublesome. With most men, the severity diminishes over time. If the hormone suppression is used for only a short period, the changes usually go back to normal when the drug is stopped and normal hormone production returns. Some men find that their sexual desire or performance is still impaired, however, especially if they have had radiation treatment simultaneously.
Men who stay on hormone suppression for a long period will experience changes in their body fat distribution which gives them a more “rounded” shape. They may be vulnerable to osteoporosis, cholesterol and diabetic problems and need to be monitored. Some men find that they have fatigue or lack of motivation that is insidious and troubling. As with most medications, LHRH drugs have their good sides and bad. Discuss your particular situation with your doctor.
Surgery
Surgery can be done to remove the prostate gland entirely, along with some or all of the seminal vesicles and, sometimes, sampling or removal of the nearby lymph nodes. After surgery, patients generally need to leave a catheter in place, usually for a matter of weeks, until the patient regains has his ability to be continent of urine. Many men regain excellent continence, though some do find that they are more likely after surgery to leak a little urine when they cough, strain, bend over, or change position, and therefore they wear an absorbent pad in their underwear.
Many men will be impotent (unable to have erections) immediately after a prostatectomy. Many years ago, this was always a permanent condition. Now it is possible to spare the nerves controlling erections, if the disease is not extensive, and while some men still never regain erections after surgery, many men get back their sexual function after some weeks or months. Many often find that they now need potency-enhancing drugs such as Viagra.
If you have a prostatectomy, be sure to ask your surgeon afterward about the stage of your tumor. If you had a T3 tumor, which means there was cancer extension outside the prostate or to the seminal vesicle, you should have radiation therapy after surgery. This is true even if the surgeon was able to get all of the cancer with a clear margin, and even if your postoperative PSA falls to zero. Two important studies, one from the US and another from Germany, have proved that the survival rate after prostatectomy for T3 disease is higher if radiation is added, but not all urologists are yet convinced. At the very least, you should have a consultation with a radiation oncologist.
External Beam Radiation Therapy (EBRT)
In this treatment high-energy radiation beams are aimed at the target area (the prostate, seminal vesicles, and surrounding tissues) from multiple directions. Treatments are given five days a week for about eight weeks. The treatment itself takes only a few minutes, produces no sensation at all, and produces no immediate side effects. There is no hair loss (other than possible partial loss of pubic hair), no burning, no sickness, and only some patients have fatigue. You do not become radioactive. There is no need to have someone else drive to and from treatments. The side effects arise from the fact that the prostate is actually in contact with the bladder and the front wall of the rectum. Because the radiation beam passes through the body and out the other side, the bladder and rectum cannot be entirely shielded from the radiation. Most of the technological progress in external beam treatment has been in providing relative sparing of normal tissues, particularly the rectum. But almost all men, as they progress through the weeks of treatment, will experience a gradual increase in the frequency of urination and bowel movements, some urgency symptoms, and some discomfort. For most men, even at their peak, these symptoms are more an annoyance and inconvenience than a real disability. About 90% of men find that these symptoms have disappeared by about 6-8 weeks after treatment is concluded.
is the result of the ever-more-powerful technology that have come into use in the past few years, plus special linear accelerator technology. With intensity-modulation, it is possible to increase the radiation dose to the target area while protecting adjacent tissues (such as the rectum) that are vulnerable to radiation damage. This allows us to give a higher radiation dose to the prostate, increasing the likelihood of cure, while limiting the dose to the rectum to reduce complications.
utilizing IMRT allows us to shorten the standard length of treatment (generally eight to nine weeks) to four to six weeks by delivering slightly higher doses of radiation during each daily treatment.
allows treatment to be delivered over five or fewer fractions at a significantly higher dose per fraction.
Proton Therapy
Proton therapy has the unique property of delivering the majority of their energy or “dose” directly in the tumor, without going beyond it, into normal tissues. This special behavior allows tumors to receive more targeted doses, while reducing damage to the healthy tissues that surround the tumor, decreasing side effects. Similar to standard radiotherapy, proton therapy is an outpatient treatment given daily over a series of weeks. Your doctor will work with you to determine whether you are a good candidate for proton therapy.
Brachytherapy
Radioactive Seed Implant
The implantation of radioactive seeds into the prostate gland allows us to treat the cancer at “short range”, from within. The “seeds” are tiny stainless-steel capsules, small enough to pass through a needle. When their radioactivity is expended, after a few months, they sit in the prostate for the rest of your life.
The radiation from a brachytherapy procedure is effectively confined to the prostate. Therefore the expected side effects are urinary: frequency, urgency, and burning with urination. There are seldom any rectal or other effects.
You will have detectable radioactivity in your body for as much as a year, but this is of such a low level that it basically presents no risk. Although you can have intercourse, we ask that you wear a condom for the first few weeks so that there is no chance of transferring a radioactive seed to your partner. For about four months, you should not have prolonged, close contact with babies, children, or pregnant women. Your radiation team will give you more specific information.
High Dose Rate Brachytherapy
This is another brachytherapy technique, using only temporary implantation of the radioactive seeds. It does not mean that a higher radiation dose is given to the prostate, but rather that the radiation is deposited at a higher rate. The preliminary measurements and the operative procedures are similar to the permanent-implant brachytherapy above, but instead of implanting the seeds themselves, multiple hollow tubes are inserted into the prostate gland. A radioactive source will travel through each of these tubes to deliver radiation to the prostate. Once the treatment is complete, the patient will not be radioactive and will not need to take special precautions around other people.
Effective, safe and compassionate patient care is our top priority
Possible Side Effects During/After Treatment:
- Side effects of radiation therapy are limited to the area that is receiving treatment.
- Side effects can include fatigue, urinary frequency, urinary urgency, burning on urination, urinary stricture, bowel urgency, loose stools/diarrhea, bleeding with bowel movements, and erectile dysfunction
- Side effects are different for each patient. Medications may be prescribed to make you as comfortable as possible.
Follow-Up Care and PSA Monitoring
Prostate cancer is almost unique among human cancers, in having a very specific and sensitive, simple blood test for followup. There is no such thing as a blood test that proves that you’re cured, but a PSA that drops after treatment and stays low for the rest of your life is the next best thing. In almost every case, a check of the PSA every six months is all that is necessary.
Whether you have a prostatectomy, EBRT, or brachytherapy, your PSA will fall after treatment. There is no doubt at all that almost all of your prostate cancer has been removed or killed. The question is whether the very last cancer cell is gone — and there is no test for that except the test of time. Your PSA needs to be followed.
After a prostatectomy, there should be no prostate cells left in the body, and therefore no source of PSA. The PSA level falls quickly, within a couple of months, to a level that is undetectable, generally considered as less than 0.01. Any detectable PSA after prostatectomy is a reason to be evaluated for further treatment.
After radiation therapy by either method, there is still a prostate gland present, so there will still be normal prostate cells that are a source of some PSA. The PSA level falls more slowly, taking perhaps six months after brachytherapy and up to a year after EBRT to reach its lowest level, and it does not fall to undetectable levels. In this case, the absolute value is much less important than the pattern, and as long as PSA levels are steadily low, we place less emphasis on the absolute value.
Effective, safe and compassionate patient care is our top priority
Latest News
Dr. Lonika Majithia publishes on The Clinical Utility of DCISionRT on Radiation Therapy Decision Making in Patients with Ductal Carcinoma In Situ Following Breast-Conserving Surgery in Annals of Surgical Oncology
Abstract Background The role of radiation therapy (RT) following breast-conserving surgery (BCS) in ductal carcinoma in situ (DCIS) remains controversial. Trials have not identified a low-risk cohort, based on clinicopathologic features, who do not benefit from RT. A biosignature (DCISionRT®) that evaluates recurrence risk has been developed and validated. We evaluated the impact of DCISionRT […]
Dr. Avani Rao publishes on plan quality effects of proton therapy for central nervous system and skull base tumors in Radiotherapy and Oncology
Abstract Purpose With reports of CNS toxicity in patients treated with proton therapy at doses lower than would be expected based on photon data, it has been proposed that heavy monitor unit (MU) weighting of pencil beam scanning (PBS) proton therapy spots may potentially increase the risk of toxicity. We evaluated the impact of maximum […]
Dr. Daniel Kim publishes on the implications of practice consolidation among radiation oncologists
Abstract Purpose Health care practices across the United States have been consolidating in response to various market forces. The degree of practice consolidation varies widely across specialties but has not been well studied within radiation oncology. This study used Medicare data to characterize the extent of practice consolidation among radiation oncologists and to investigate associated […]
Cancer Types
Radiation Oncology Associates is the leader in comprehensive and cutting-edge radiation oncology therapy services in convenient and accessible locations across Northern Virginia.